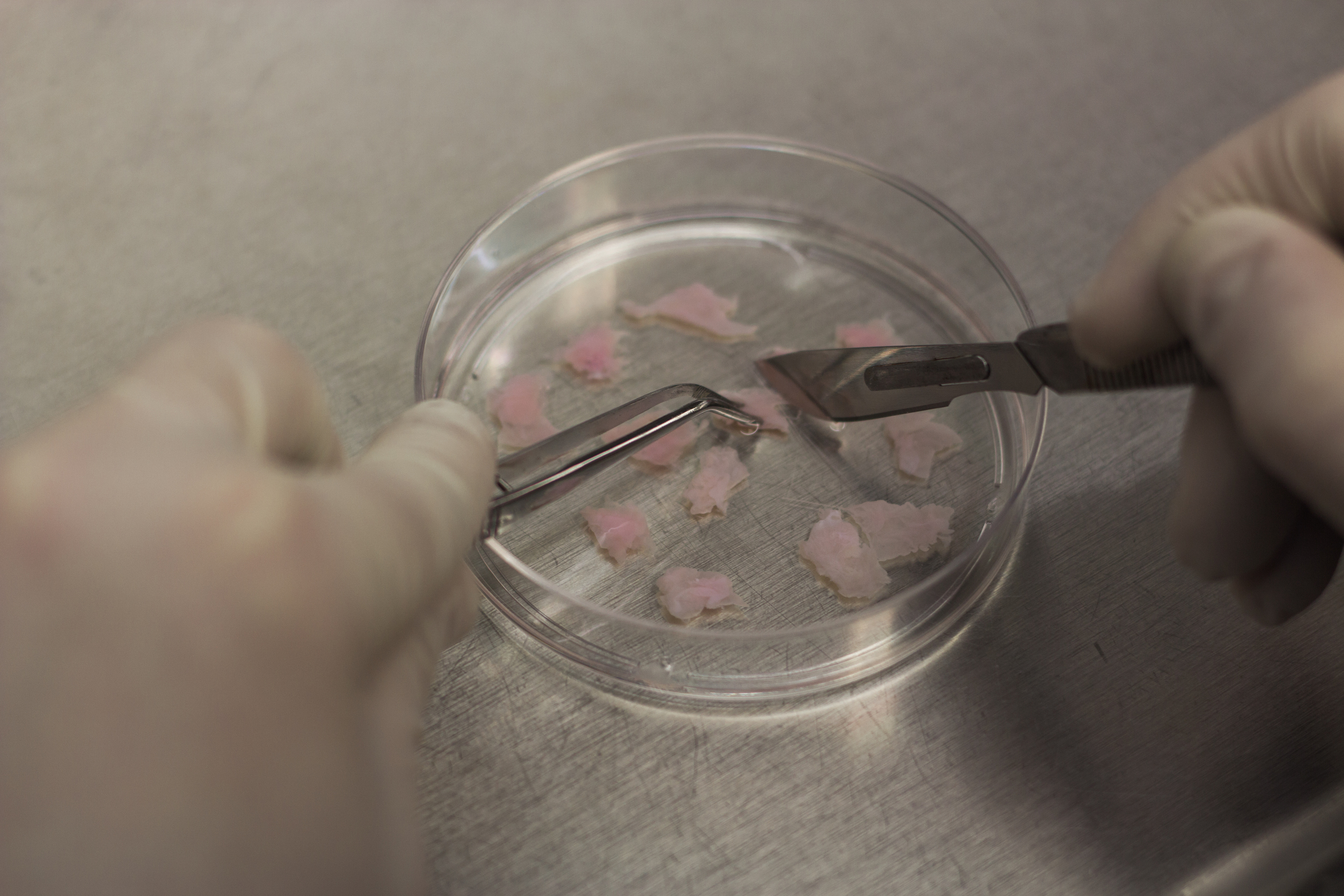
characterization of Wear Particles
Tissue wear naturally occurs as the result of the interaction between two distinct moving surfaces. Increased cartilage wear, leading to formation of loose particles in the joint space, has been linked to aging, overuse and obesity, often leading OA. Clinically, cartilage and bone debris are bound to the surface or embedded deep within the synovium in OA patients suffering from capsular synovial hyperplasia and metaplasia. Despite mounting in vitro and in vivo evidence over more than six decades of the deleterious effects of cartilage particulates on joint health, the mechanisms that mediate their negative interactions with the cells of the synovium remain poorly understood. As opposed to non-biologic (metal or plastic) wear particles, biologic (cartilage) wear particles are composed of extracellular matrix constituents that are degradable and may interact with the cell surface in addition to being internalized, and therefore are inherently more complex to study. In contrast to past studies of wear particles generated naturally in the joint or tribological wear studies, we have recently had success in performing precise quantitative analyses of particulates using a Coulter counter. Utilizing both bovine explants and a tissue engineered synovium model with clinical human cell sources, we are expanding upon this research to take tissue-level engineering measurements, characterizing synovium tribology in normal and osteoarthritic conditions.
Tissue engineering Synovium
The synovium is a thin layer of tissue comprised of macrophages and fibroblast-like synoviocytes (FLS) that lines the non-cartilaginous surfaces of articular joints. Within the tissue, synovial fibroblasts are responsible for maintaining the synovial architecture and ECM as well as synthesizing the lubricating molecules that make up synovial fluid. The pathologic thickening of the synovial joint has been attributed to the response of synovial fibroblasts to pro-inflammatory stimuli. Additionally, in OA the concentration of lubricating molecules synthesized by synovial fibroblasts is significantly decreased. Using a collagen gel scaffold model, we have developed an in vitro model for tissue engineering the synovium. This platform enables us to study how pro-inflammatory factors modulate the composition of the synovium, behavior of synovial fibroblasts, and transport across this important membrane. Further, this system can be adapted to a co-culture model to elucidate the mechanisms dictating the interaction between cartilage and the synovium in the pathologic joint.
3D Cell Migration for Injury Modeling
We have demonstrated galvanotaxis, or electric field-induced directed migration, of various cell types using a 2D galvanotaxis system, including stem cells and chondrocytes. Recently, we developed a system for examining the migration of these cells in three dimensions. Applied electric fields can be applied in vitro to foster development of functional tissue grafts in culture as well as in vivo for promoting tissue repair. The application of electric fields is already used clinically to promote wound healing of various tissues, including skin, bone and cartilage. Application of DC electric fields will expedite migration of reparative cells that can be delivered either intra-articularly or from the synovial lining of the joint, as modeled using our tissue engineered synovium. Modulation of the field to dynamic fields can then modulate biosynthetic capacity of the cells. Electric fields can therefore modulate cell migration, orientation, as well as biosynthetic activities.